Bizarre Boiling
- by NASA Marshall Space Flight Center and ScienceIQ.com
 The next time you're watching a pot of water boil, perhaps for coffee or a cup of soup, pause for a moment and consider: what would this look like in space? Would the turbulent bubbles rise or fall? And how big would they be? Would the liquid stay in the pan at all? Until a few years ago, nobody knew. Indeed, physicists have trouble understanding the complex behavior of boiling fluids here on Earth. Perhaps boiling in space would prove even more baffling.... It's an important question because boiling happens not only in coffee pots, but also in power plants and spacecraft cooling systems. Engineers need to know how boiling works.
The next time you're watching a pot of water boil, perhaps for coffee or a cup of soup, pause for a moment and consider: what would this look like in space? Would the turbulent bubbles rise or fall? And how big would they be? Would the liquid stay in the pan at all? Until a few years ago, nobody knew. Indeed, physicists have trouble understanding the complex behavior of boiling fluids here on Earth. Perhaps boiling in space would prove even more baffling.... It's an important question because boiling happens not only in coffee pots, but also in power plants and spacecraft cooling systems. Engineers need to know how boiling works.
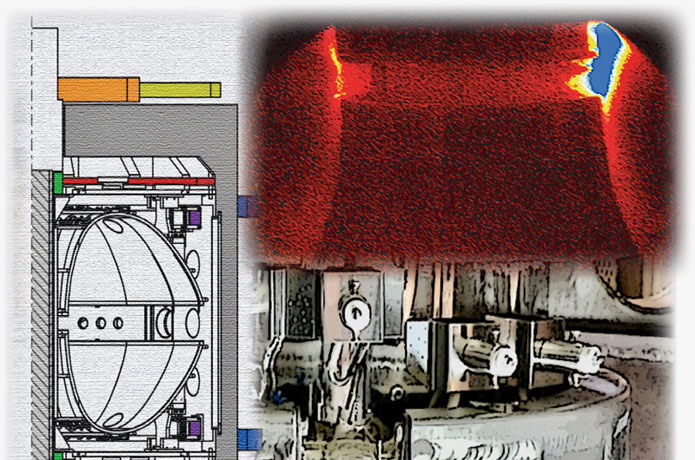
In the early 1990's a team of scientists and engineers from the University of Michigan and NASA decided to find out. Using a freon coolant as their liquid, they conducted a series of boiling experiments on the space shuttle during 5 missions between 1992 to 1996. And indeed, they found some intriguing differences between what happens to boiling fluids on Earth and what happens to them in orbit. For example, a liquid boiling in weightlessness produces -- not thousands of effervescing bubbles -- but one giant undulating bubble that swallows up smaller ones!
Despite its entertainment value, this research is much more than a simple curiosity. Learning how liquids boil in space will lead to more efficient cooling systems for spacecraft, such as the ammonia-based system on the International Space Station. Knowledge of boiling in space might also be used someday to design power plants for space stations that use sunlight to boil a liquid to create vapor, which would then turn a turbine to produce electricity.