Flashing LED Circuit DIY Electronics Kit
$4.99$2.95
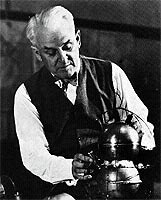 The credit goes to a Professor of Physics at the University of Chicago Robert Andrews Millikan (1923 Nobel Prize Winner in Physics). He determined the charge on an electron by causing electron laden oil-drops (thus the name 'charged oil-drop experiment) to fall between two electrically charged plates (the top one +ve, bottom -ve). he then repeated the experiment, but this time with uncharged plates.
By measuring the difference in how fast they fell, he was able to calculate the charge on an oil drop. (He did this by equating Coulomb's law to Newton equations of motion). After repeating this experiment several times, he deduced that all his answers were whole number multiples of 1.6 * 10-19 Coulombs (that's a decimal place followed by 18 zeros and then 16). He thus deduced that the charge on one electron was 1.6*10-19 C. He was also able to deduce the mass of an electron using the charge to mass ratio.
The credit goes to a Professor of Physics at the University of Chicago Robert Andrews Millikan (1923 Nobel Prize Winner in Physics). He determined the charge on an electron by causing electron laden oil-drops (thus the name 'charged oil-drop experiment) to fall between two electrically charged plates (the top one +ve, bottom -ve). he then repeated the experiment, but this time with uncharged plates.
By measuring the difference in how fast they fell, he was able to calculate the charge on an oil drop. (He did this by equating Coulomb's law to Newton equations of motion). After repeating this experiment several times, he deduced that all his answers were whole number multiples of 1.6 * 10-19 Coulombs (that's a decimal place followed by 18 zeros and then 16). He thus deduced that the charge on one electron was 1.6*10-19 C. He was also able to deduce the mass of an electron using the charge to mass ratio.
 'There is no inductive method which could lead to the fundamental concepts of physics. Failure to understand this fact constituted the basic philosophical error of so many investigators of the nineteenth century.'
'There is no inductive method which could lead to the fundamental concepts of physics. Failure to understand this fact constituted the basic philosophical error of so many investigators of the nineteenth century.'